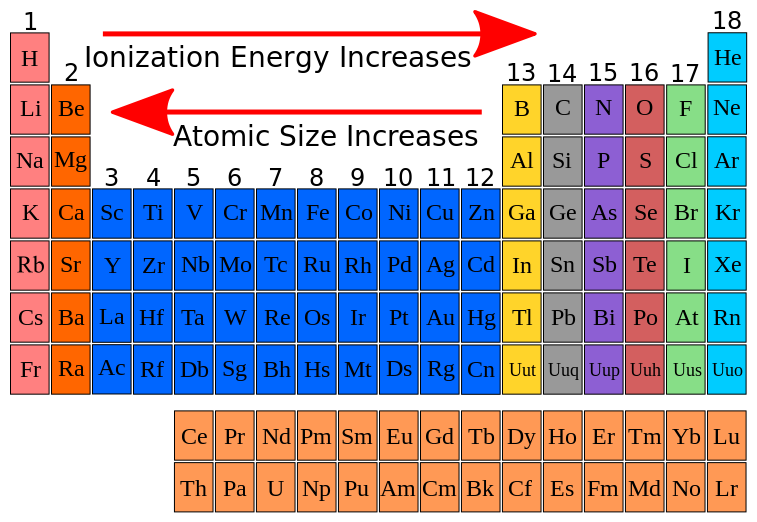
Remember these guys over here group one and group two where we know it's relatively easy to remove an electron because they're interested in being oxidized versus our non-metals which tend to be on the right hand side of the periodic table, are going to have higher ionization energies because that's where like your halogens and your noble gases are and they're not interested in giving away electrons but in receiving them. In general, the first ionization energy increases as we go from left to right across a row of the periodic table. So knowing that, that means that metals have the lowest ionization energies. So at the top right hand corner of the periodic table is where you're going to have the highest ionization energy. So remember the periods are the rows going from left to right. And then ionization energy increases across a period. So down the columns ionization energy decreases which means towards the bottom of the periodic table is where you're going to find the lowest ionization energy meaning this is where it's the easiest to remove an electron from. So let's just make it as simple as possible and so a couple of things you're going to want to remember is that ionization energy decreases down a group on the periodic table. So the first ionization energy might be fairly low, then the second one gets a little bit higher, third one even higher and that's because the electrons are being pulled away from a more positive ion as you continue to remove electrons and that requires even more energy. So you can have a first ionization energy, second, third, fourth and so forth and so on. So ionization energy increases as successive energy electrons are removed. So, for instance, group one and two group two metals are really simple to remove the electrons versus the non-metals up on the right hand side of the periodic table will be much more difficult to remove an electron.
So basically, the greater the ionization energy the more difficult it's going to be to remove an electron.
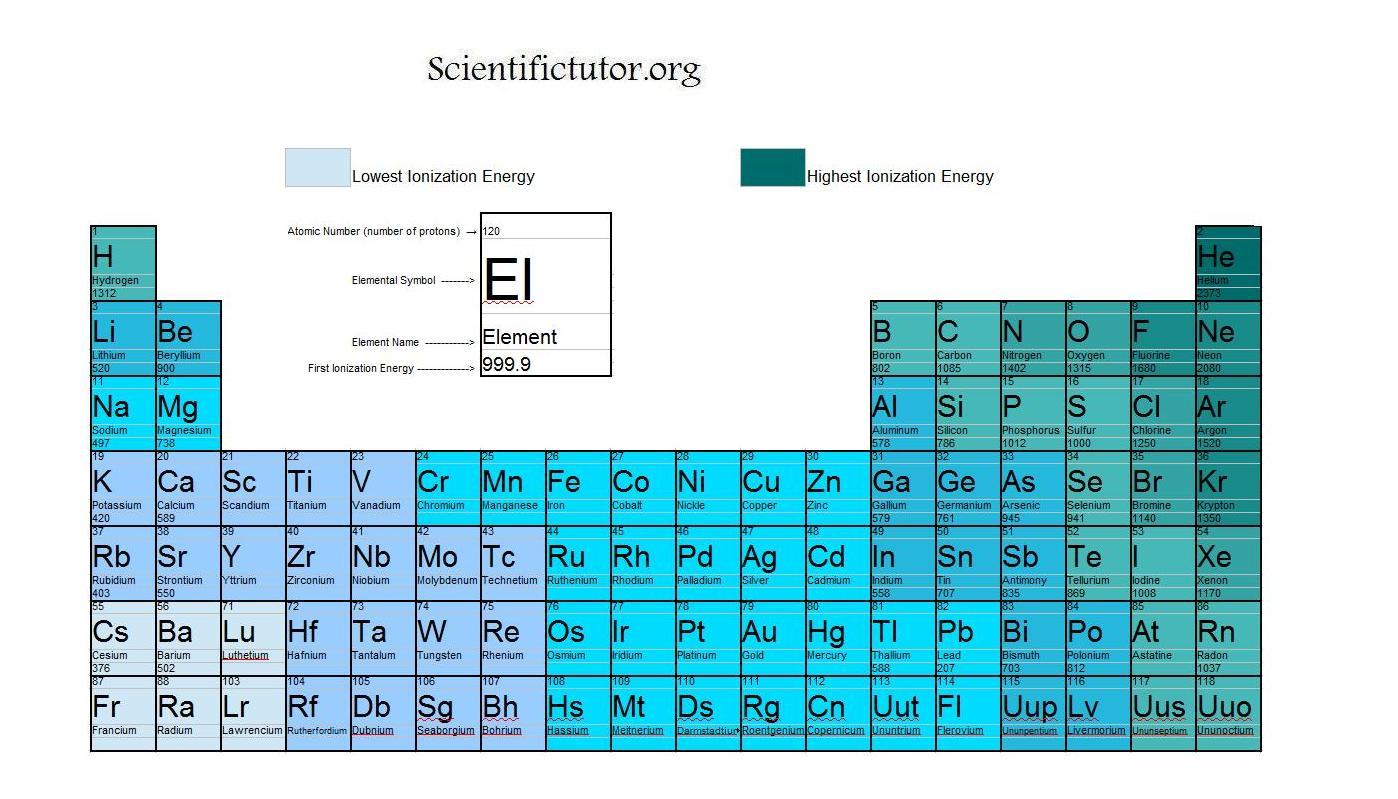
X + energy X + + e where X is any atom or molecule capable of being ionized, X + is that atom or molecule with an electron removed (positive ion), and e is the removed electron. So of course this would have an impact on the chemical behavior because this talks about how electrons move between metals and non-metals and things of that nature. Periodic Table Ionization Energy Ionization energy, also called ionization potential, is the energy necessary to remove an electron from the neutral atom. So this is the minimum energy that's required to remove an electron from the ground state. So you'll recall from kind of learning about the structure of an atom, how the ground state and an excited state of an atom or an ion work. So the definition of ionization energy is that it's the minimum energy required to remove an electron from the ground state of an atom or an ion. So let's go ahead and talk about ionization energy which is another property that kind of comes from our understanding of the periodic table.

